Key Experiment
The Experiment
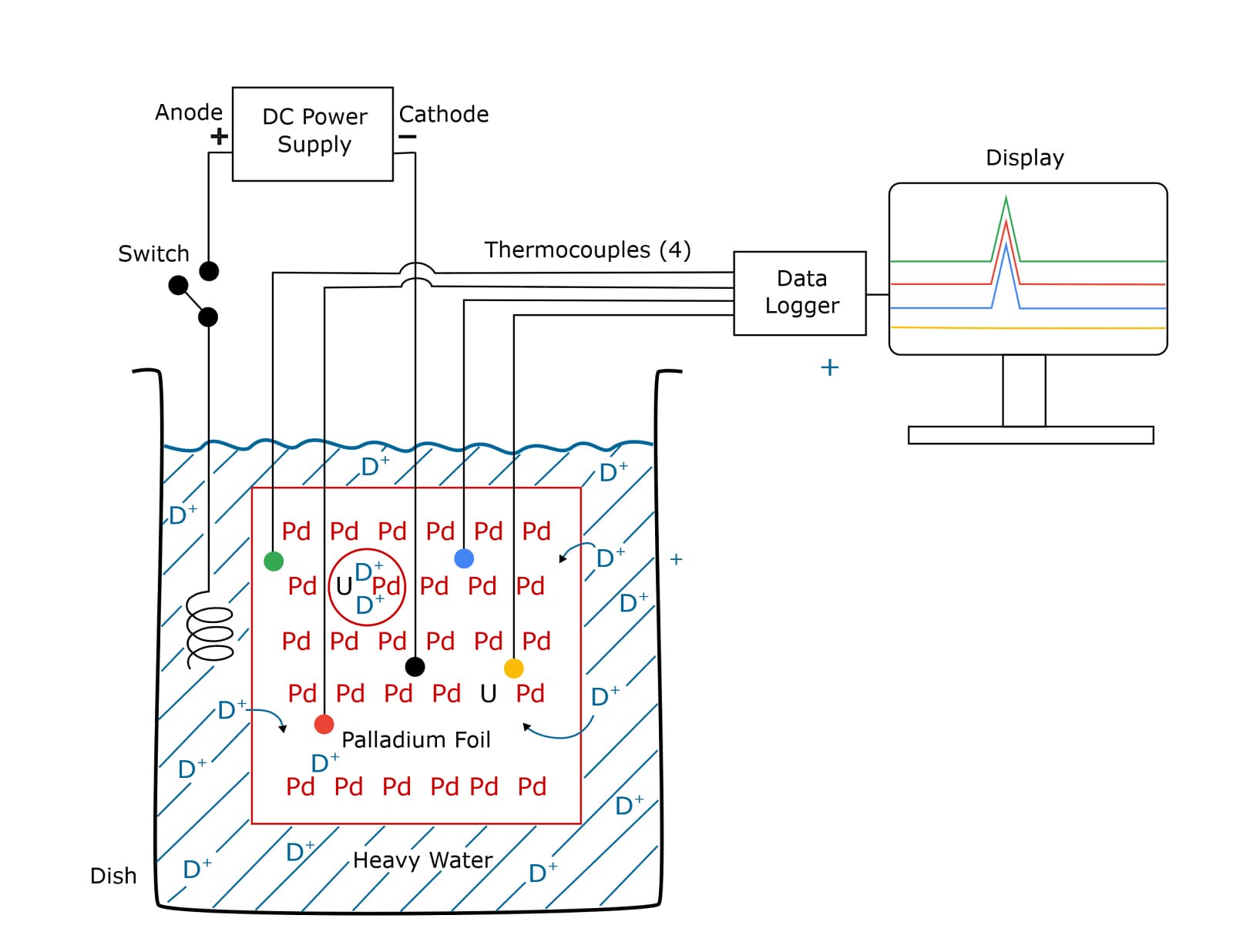
The center of the experiment was a simple electrolytic cell shown in the drawing below. This consisted of an open borosilicate dish (shown in black) filled with a concentrated electrolyte, a solution of lithium deuteroxide (LiDO) in heavy water (D2O). The cathode was a palladium (Pd)-uranium (U) alloy foil (shown in red) fully submerged into the electrolyte and attached to an insulated platinum wire that connected it to a DC power supply through a switch.
Four thermocouples are attached to the small palladium foil in four different locations. The foil was about the size and thickness of a postage stamp. The thermocouples were connected to a multi-channel data logger and their temperature outputs were displayed on a PC screen.
The temperature outputs were constant while the palladium foil was being charged with deuterium ions, D+. (See arrows showing some D+ ions moving from the electrolyte into the foil.) If a uranium atom that was close to two deuterium ions (as inside the red circle) were to spontaneously decay, sufficient heat would be produced to cause D-D fusion.
After two hours of charging, the display burst into life showing simultaneous temperature spikes had been detected by three of the four thermocouples. Minutes later, the fourth thermocouple also registered a temperature spike. Each spike had the calculated energy equivalent of billions of D-D fusion events – implying a chain reaction occurred. This signaled that the palladium-foil reactor had gone CRITICAL.
It became the smallest controlled nuclear fusion reactor ever operated. The amount of heat produced was adjusted up or down by increasing or decreasing the electrical current. This is analogous to moving the control rods in or out of a conventional fission reactor. The palladium-foil reactor was continuously operated for several hours before making a controlled shut-down.
This experiment confirmed the effect of Solid-State Fusion.
Setup Photos
Upper left: The electrolytic cell [shown schematically, in The Experiment] containing a postage-stamp-sized palladium foil (cathode -) and a serpentine platinum wire (anode+).
Lower right: Same setup as above including 3 orange spring-loaded clips used to secure the cathode lead wire (middle clip) and 4 thermocouples (left and right clips) to the palladium foil. These clips facilitate rapid removal of the foil to precisely measure its weight gain due to electrolytic loading of deuterium into the foil.